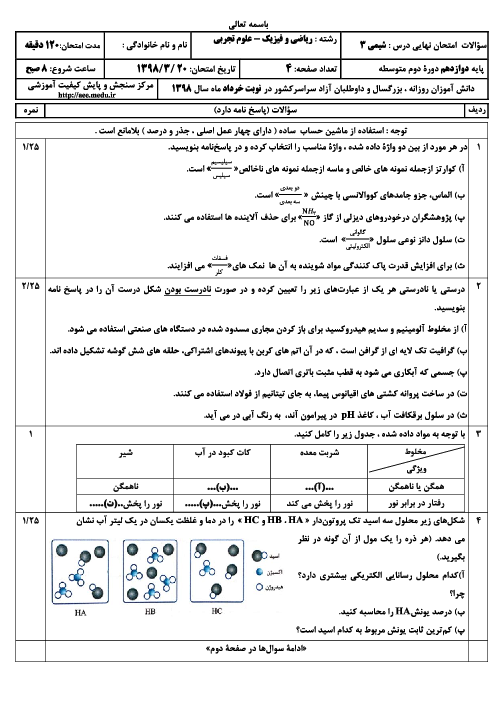
$F{{e}_{2}}{{O}_{3}}(s)+3CO(g)\rightleftharpoons 3Fe(l)+3C{{O}_{2}}(g)$ $BaO(s)+C{{O}_{2}}(g)\rightleftharpoons BaC{{O}_{3}}(s)$ $4Fe(s)+3{{O}_{2}}(g)\rightleftharpoons 2F{{e}_{2}}{{O}_{3}}(s)$ $2NO(g)\rightleftharpoons {{N}_{2}}(g)+{{O}_{2}}(g)$ $2N{{O}_{2}}(g)\rightleftharpoons {{N}_{2}}{{O}_{4}}(g)$
رابطه ثابت تعادل را برای هر یک از واکنشهای زیر نوشته و یکای آن را مشخص کنید.
پاسخ ها: {{ repliesNum }}
پاسخ انتخاب شده
در پاسخ به: {{ reply.reply_to.name }}
در پاسخ به
این پیام حذف شده است.