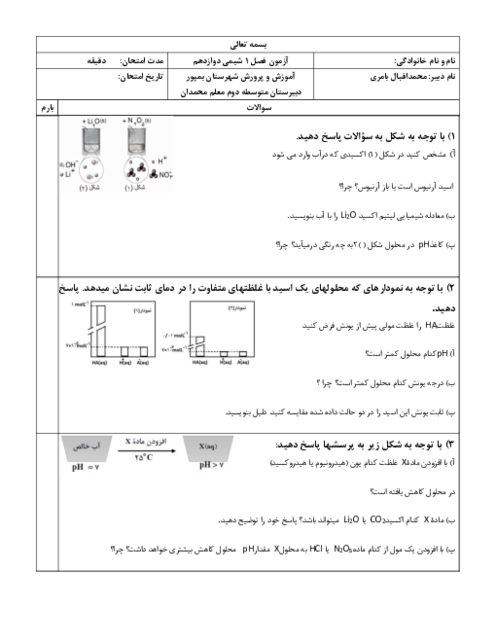
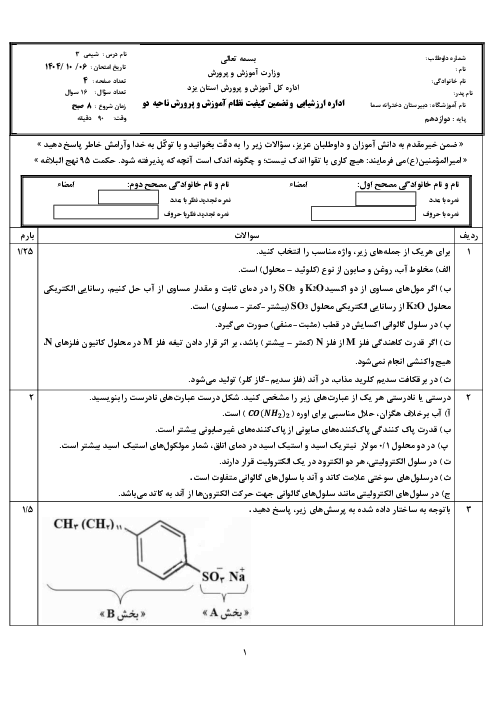
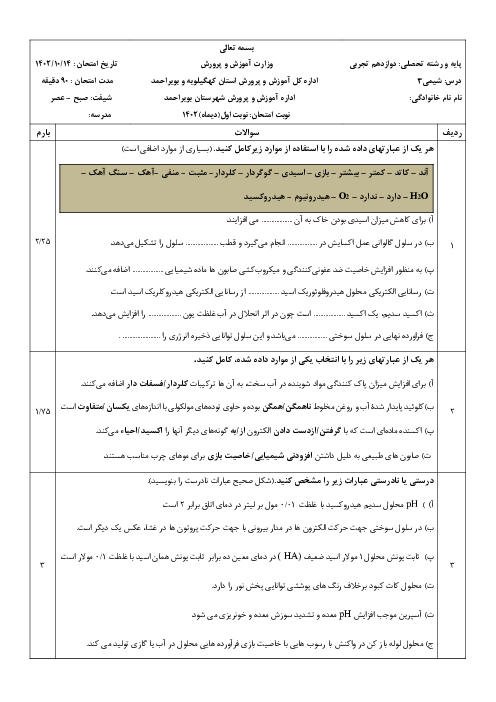
$\begin{align}
& Ba{{(OH)}_{2}}\Rightarrow \left[ {{H}^{+}} \right]={{10}^{-pH}}={{10}^{-13}}mol.{{L}^{-1}} \\
& \Rightarrow \left[ O{{H}^{-}} \right]=\frac{{{10}^{-14}}}{{{10}^{-13}}}={{10}^{-1}}mol.{{L}^{-1}} \\
& HCl\Rightarrow \left[ {{H}^{+}} \right]={{10}^{-pH}}={{10}^{-1/7}}={{10}^{-2}}\times {{10}^{0/3}}=2\times {{10}^{-2}}mol.{{L}^{-1}} \\
& \left[ O{{H}^{-}} \right]=\frac{\left| {{10}^{-1}}\times 0/1-2\times {{10}^{-2}}\times 0/15 \right|}{0/1+0/15} \\
& =\frac{0/007}{0/25}=0/028mol.{{L}^{-1}} \\
& \Rightarrow \left[ {{H}^{+}} \right]=\frac{{{10}^{-14}}}{\left[ O{{H}^{-}} \right]}=\frac{{{10}^{-11}}}{28}mol.{{L}^{-1}} \\
& pH=-\log \left| {{H}^{+}} \right|=-\log ({{10}^{-11}})-\log (\frac{1}{28})=11+\log (28) \\
& =11+\log 4+\log 7=11+0/6+0/85=12/45 \\
\end{align}$